As part of the Wild Tolworth project, the Biological Recording Company was commissioned by Citizen Zoo to run a series of seven Field Recorder Days at Tolworth Court Farm, in the London borough of Kingston, during 2024 and 2025. Thanks to funding from the National Lottery Heritage Fund, we were able to build on previous events delivered at the site in partnership with the London Natural History Society and Field Studies Council (the latter as part of the FSC BioLinks project). The Field Recorder Day events in 2024 and 2025 covered beetles, dragonflies, grasses, flies, fungi and pollinators. This blog shares some highlights from these events.

1. Pollinator Field Recorder Day
🗓️ When: 29 May 2024 | Specialist: Abigail Lowe
For our first event of 2024 we welcomed 23 Field Recorder Day participants to Tolworth Court Farm to join pollinator specialist Abigail Lowe in recording bees, hoverflies, butterflies and anything else that pollinates. It was a glorious Spring day, with lots of pollinators out and about. The most exciting finds from the day included: (1) the bumblebee-mimicking hoverfly Volucella bombylans, whose larvae live as scavengers and larval predators in the nests of bumblebees and social wasps; (2) the Green Hairstreak butterfly Callophrys rubi, one of the more uncommon butterflies which is a fan of scrubby plants and hedgerows; and (3) the Sleepy Carpenter Bee Chelostoma florisomne, so-named for the predilection of males to ‘lie down’ in buttercup flowers during overcast weather. Across the day a total of 130 biological records were generated, representing 80 species and including 11 bee species, 11 hoverflies and 9 butterflies. A big thanks to everyone who came along and helped out.


2. Grasses, Sedges and Rushes Field Recorder Day
🗓️ When: 14 June 2024 | Specialist: Mark Spencer
In June 2024, 13 attendees joined us and botanist Mark Spencer for a Field Recorder Day focused on grasses, sedges and rushes. Generally considered more difficult to identify than flowering herbs, shrubs or trees, these plants often fall to the wayside when it comes to botanical recording. A targeted Field Recorder Day was therefore called for, and indeed was especially warranted given the dominant habitat at Tolworth Court Farm is grassland. Interesting finds from the day included: (1) Meadow Fescue Festuca pratensis, an uncommonly recorded grass in London, which favours neutral grasslands; (2) Slender Soft-brome Bromus lepidus, a non-native neophyte spread as a contaminant of grass seed mixtures; and (3) False Fox-sedge Carex otrubae, which we found growing extensively in a damp corner of Tolworth Court Farm Fields. In total, across the day, 81 records were generated of 34 species including 23 grass species, 2 sedges and 1 rush.

3. Dragonfly Field Recorder Day
🗓️ When: 23 July 2024 | Specialist: Linda Pryke
In July 2024, 16 attendees joined us and dragonfly specialist Linda Pryke for a day surveying the dragonflies and damselflies of Tolworth Court Farm. Given the site is situated along the banks of the Hogsmill river, one of London’s precious few chalk streams, there is potential habitat for these beautiful insects. Across the day we recorded four species of damselfly – Banded Demoiselle Calopteryx splendens, Beautiful Demoiselle Calopteryx virgo, Large Red Damselfly Pyrrhosoma nymphula and Blue-tailed Damselfly Ischnura elegans – but no dragonflies. The highlight was definitely Calopteryx virgo, a scarce species in London which seemingly maintains its stronghold in rivers and wetlands in the southwest of the city. In total across the day we generated 32 records of 19 species including the 4 damselflies.

As of Autumn 2025, Citizen Zoo have begun construction of a new wetland area at Tolworth Court Farm Fields. This hopefully means more dragonflies and damselflies in the future!
4. Fungi Field Recorder Day
🗓️ When: 9 October 2024 | Specialist: Mark Spencer
As summer turned to autumn we changed our focus away from botany and entomology and turned instead to fungi. Autumn is the best season for surveying fungi, as the wetter weather and decaying vegetation creates damp conditions in which fungi thrive. In October we were back at the site with Mark Spencer (who does mycology as well as botany) and 29 enthusiastic Field Recorder Day participants, ready to search for fungi. Over the course of the day we found 16 different species and generated 31 records. Some of the species found included: (1) the Jelly Ear fungus Auricularia auricula-judae found on deadwood; (2) the attractively ‘white-dusted’ Cortinarius hemitrichus (Frosty Webcap); and (3) the uncommonly recorded Hebeloma sacchariolens (Sweet Poisonpie). A very nice bonus was the tiny fungus-feeding beetle Agaricochara latissima (Staphylinidae) which was found on the fungus Daedaleopsis confragosa – this was a first record of the species for Greater London on iRecord!


5. Beetle Field Recorder Day
🗓️ When: 25 April 2025 | Specialist: Connor Butler
Our first event of 2025 was a Beetle Field Recorder Day. We invited beetle specialist Connor Butler to lead the day and had 24 participants. This event was by far our most productive of the project so far, with an impressive 273 total records generated. These records covered 141 different species including 54 different beetles. Highlights among these included (1) the weevils Oxystoma craccae and Oxystoma pomonae – distinctive for their strange ‘wedge-shaped’ rostrums – which feed on Vicia sativa (Common Vetch); (2) Elaphrus riparius, a ground beetle (family: Carabidae) restricted to the margins of freshwater; and (3) the click beetle Agriotes lineatus (Elateridae) which was photographed particularly nicely by Mick Massie (see right). If you want to read more about what we got up to check out our blog summarising the day.


6. Fly Field Recorder Day
🗓️ When: 22 July 2025 | Specialist: Martin Harvey
Containing around 7,100 known UK species, the order Diptera (true flies) makes up a considerable proportion of British animal diversity. On a prime July day we returned to Tolworth under the guidance of Diptera specialist Martin Harvey to see which flies we could find on the site. Despite a lower number of participants than other events (8), the team was very productive, generating 111 records and recording 39 fly species. Very excitingly, one of these species was a first record for Greater London as per iRecord and the NBN Atlas: Chamaemyia juncorum, a member of a small family of British flies, the Chamaemyiidae, which are predators of aphids. A further two species found on the day were second records for Greater London: Helina obscurata (Muscidae) and Scatella stagnalis (Ephydridae). Besides the rarities, good numbers of some more common species were also seen, including 12 different hoverfly species.

7. Fungi Field Recorder Day
🗓️ When: 15 October 2025 | Specialist: Mark Spencer
For our final event of the project, we ran a second Field Recorder Day focused on Fungi, again utilising the ever-wonderful Mark Spencer as our specialist. Keen to add to the species list generated last time, we were pleased to welcome a record-breaking 40 attendees to the site to search high and low for mushrooms and their kin. And add to the species list we did indeed, with the team collectively finding and identifying 39 different fungi species, 33 of which we did not find on our first visit. Interestingly, this also means 6 of the 16 species found in 2024 were not re-found in 2025. More fungi surveying is clearly warranted – there are probably many more species to be recorded on site. Some of the fungi species found on this visit but not the previous visit were the mushrooms Bolbitius titubans, Coprinellus disseminatus and Entoloma sericeum, the crust fungi Dichomitus campestris and Kretzschmaria deusta and the powdery mildews Erysiphe heraclei and Erysiphe trifoliorum. Read more about what we got up to on this day in our Recording London’s Fungi blog.


Project Achievements
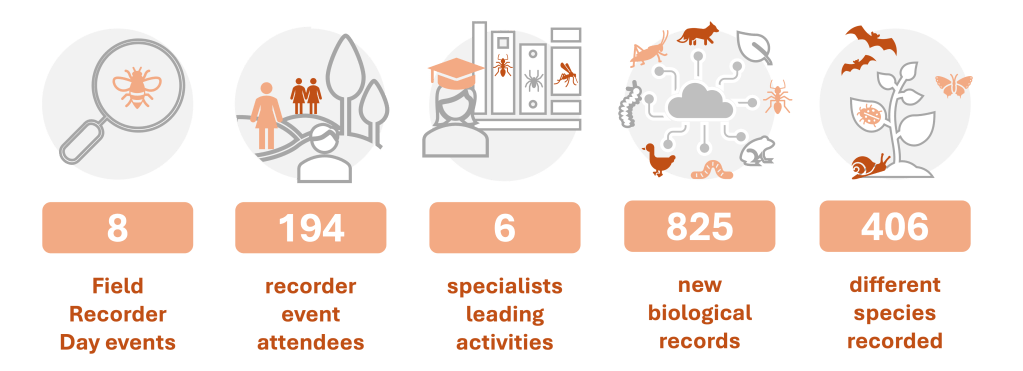
Across the seven events summarised above, plus one additional Invertebrate Field Recorder Day ran in 2023 at Tolworth Court Farm as a collaboration between Biological Recording Company and the London Natural History Society (not formally part of the contract but grouped with it for reporting purposes), 186 people were engaged in biological recording, 825 new biological records were generated and 406 different species were recorded. The vast majority of the species found represent first records for Tolworth Court Farm. All records contribute towards building a picture of the biodiversity present on site and will inform future conservation management work.
As always, our data is gathered through iRecord so it is automatically accessible to Greenspace Information for Greater London (GiGL) and the relevant National Recording Schemes & Societies.
You can read the full report covering all of our recording activities and achievements at Tolworth Court Farm below:
Thank you very much to all who attended and and contributed valuable data to the project. A big thanks also to Elliot Newton and the Citizen Zoo team for helping to deliver these events, and to our specialists Abi Lowe, Mark Spencer, Linda Pryke, Connor Butler and Martin Harvey for their knowledge and guidance. Finally, thank you to the London Natural History Society for helping to promote the events.We hope to see you at future events!
The above series of events was delivered by the Biological Recording Company in partnership with Citizen Zoo and Kingston Council as part of the Wild Tolworth project (supported by the National Lottery Heritage Fund).

Biological Recording Projects
At the Biological Recording Company, we specialise in planning and delivering projects centred around recording wildlife and training naturalists.
- London Recording Projects involve the delivery of events from our Field Recorder Day, Invertebrate Study Day, Earthworm Sampling Day and Training Course programmes. Examples include:
- Site-based projects, such as Wild Tolworth, Ealing Beaver Project and Lesnes 500.
- Borough-based projects, such as our Southwark and Barnet projects.
- Earthworm Projects range from identification training and earthworm surveys to research and consultation. Example include:
- Engagement-focused projects, such as delivering earthworm recorder training in Northern Ireland and working with farmers in the Chilterns.
- Research-focused projects, such as looking at the impact of hay meadow restoration on earthworm communities, assessing the effectiveness of regenerative farming practices and investigating the use of AI in earthworm identification.
- National Biological Recording Projects entail putting our expertise to use with helping other organisations improve the biological recording processes and reach new audiences.
- Training projects include developing online training for Lepidoptera verifiers and producing guidance on the use of Record Cleaner and iRecord.
- Consultation projects include reviewing the pay of natural history tutors and working with the British Mycological Society to improve fungus record quality and data flow.