The Biological Recording Company is working with a range of organisations, including Barnet Council and Citizen Zoo, to deliver a programme of free natural history training courses and biological recording events throughout 2025 and 2026, including:
- Online ecology and biodiversity courses that can be completed at your own pace.
- Field Recorder Day events to record the wildlife of the Welsh Harp under the guidance of biodiversity specialists.
- In-person natural history training courses across sites in Barnet.
More details about the programme, including links to the booking pages for the various activities, can be found below.
This series of training events in Barnet is being run as a collaboration between the Biological Recording Company and Barnet Council.

Online Training Courses
Barnet residents and volunteers can access any of our online courses and virtual symposium content for free. We have 200 spaces to give away so get in touch with Harriet at Barnet Council for instructions on how to claim your free courses. A full list of eligible courses is provided below.
entoLEARN online courses
- Bee-flies of the UK
- Bumblebees of the UK
- Chafers of the UK
- Damselflies of the UK
- Dragonflies of the UK
- Earthworms of the UK and Ireland
- Freshwater Leeches of the UK
- Harvestmen of the UK
- Longhorn Beetles of the UK
- Social Wasps of the UK
- Soldierflies of the UK
Skills For Ecology online courses
- Biodiversity and the UN Sustainable Development Goals
- Camera Traps
- Crayfish Surveys and Conservation
- Drone Surveys for Ecology
- Ferns of the UK
- Flower-Insect Timed (FIT) Counts
- Hay Meadow Restoration
- Identifying Mosses
- Invasive Plants in the UK
- iRecord 101
- Surveying For Beetles
- Surveying For Dragonflies
- Surveying For Pollinators
- Wildlife Detection Dogs
Virtual Symposium Content
- Beetle Research and Conservation
- Biological recording
- Funding For Conservation
- Hedgehog Research
- Invertebrate Translocation and Reintroduction
- Rewild London
- Wildlife Gardening
Instructions for claiming courses funded by Barnet Council
- Navigate to the relevant product on the Biological Recording Company online learning platform: https://courses.biologicalrecording.co.uk/collections/products
- Click on the button ‘Buy £X‘ to bring up the basket page.
- Click on the text stating ‘Have a coupon?‘ to bring up the coupon box
- Enter the code provided by Barnet Council into the coupon box and click ‘Apply‘ to reduce the cost to £0. Barnet residents and volunteers should contact Harriet at Barnet Council to request a code.
- Enter your email address, first name and last name to complete the purchase and set up your account.
Field Recorder Days
Following on from the training, we’ll be putting our existing and newly-developed skills to good use at the Welsh Harp Open Space with a series of Field Recorder Day events. Each event is led by a species group specialist and will have a specific taxonomic focus. Join us to help record the wildlife of the Welsh Harp.
- Earthworm Sampling Day 01 Apr 2026 at Welsh Harp (Barnet)
- Invertebrate Field Recorder Day 29 May 2026 at Welsh Harp (Barnet)
- Botany Field Recorder Day 24 Jun 2026 at Welsh Harp (Barnet)
- Plant Gall Field Recorder Day 19 Sep 2026 at Welsh Harp (Barnet)
- Fungi Field Recorder Day 15 Oct 2026 at Welsh Harp (Barnet)
In-person Training Courses
Eight one-day natural history training courses are scheduled at sites within the London Borough of Barnet, with subjects aimed at those new to biological recording and an emphasis on developing biological recording skills in Barnet residents and greenspace volunteers.
Upcoming courses are listed below (previous courses are reported on in the Project Achievements section below). All courses are free, but spaces are limited, so we recommend booking early to avoid disappointment.
Urban Grassland Management coming soon…
Project Achievements
In-person Training Courses

Biological Recording 101 took place at Hendon Town Hall on 3 June 2025, taught by Keiron Brown and attended by 16 participants. The course highlighted the value of biological recording for understanding and protecting wildlife. Attendees learned to collect and submit species data via iRecord and manage records to support local biodiversity monitoring.
Teaching Natural History took place at Barnet Environment Centre on 8 January 2026, taught by Keiron Brown and attended by 9 participants. Attendees learnt how to confidently teach adults through engaging classroom and field-based learning, and gained practical tools to design, deliver, and evaluate effective natural history education.
Fungi For Beginners took place at Hendon Town Hall on 15 January 2026, taught by Mark Spencer and attended by 14 participants. Attendees were introduced to the fascinating world of British fungi and learnt the basics of finding and identifying them through classroom learning and guided fieldwork. Participants gained foundational skills in fungal biology, taxonomy, and safe field practice.
Winter Tree Identification took place at Barnet Environment Centre on 28 January 2026, taught by Henry Miller and attended by 14 participants. Attendees learnt how to identify broadleaved trees in winter using buds, twigs, bark, and seasonal features, and gained confidence through practical exercises and guided fieldwork.
Botany For Beginners
03 Feb 2026 @ Hendon Town Hall
Discover British plants and gain practical skills in identifying them, recognising key structures, and understanding their taxonomy and ecology.
Camera Trapping Mammals
18 Feb 2026 @ Freehold Community Centre
Master camera trapping to monitor British mammals and classify footage using MammalWeb. Gain hands-on experience setting up cameras, identifying species, and managing data.
Terrestrial Invertebrates for Beginners
26 Mar 2026 @ Golders Green Quaker Meeting House
Explore British terrestrial invertebrates and develop core skills in finding and identifying them through guided fieldwork. Gain confidence using identification keys and recognising key features.
Virtual Events

The Wildlife Gardening Virtual Symposium took place on 14th January 2026, and 47 free spaces were claimed by Barnet residents/volunteers. Researchers and practitioners explored how gardens and urban green spaces support wildlife, highlighting ponds, pollinator-friendly gardening, fungi, and urban mammals through presentations on habitat creation, ecological interactions, and long-term citizen science research.
Biological Recording Projects
At the Biological Recording Company, we specialise in planning and delivering projects centred around recording wildlife and training naturalists.
- London Recording Projects involve the delivery of events from our Field Recorder Day, Invertebrate Study Day, Earthworm Sampling Day and Training Course programmes. Examples include:
- Site-based projects, such as Wild Tolworth, Ealing Beaver Project and Lesnes 500.
- Borough-based projects, such as our Southwark and Barnet projects.
- Earthworm Projects range from identification training and earthworm surveys to research and consultation. Example include:
- Engagement-focused projects, such as delivering earthworm recorder training in Northern Ireland and working with farmers in the Chilterns.
- Research-focused projects, such as looking at the impact of hay meadow restoration on earthworm communities, assessing the effectiveness of regenerative farming practices and investigating the use of AI in earthworm identification.
- National Biological Recording Projects entail putting our expertise to use with helping other organisations improve the biological recording processes and reach new audiences.
- Training projects include developing online training for Lepidoptera verifiers and producing guidance on the use of Record Cleaner and iRecord.
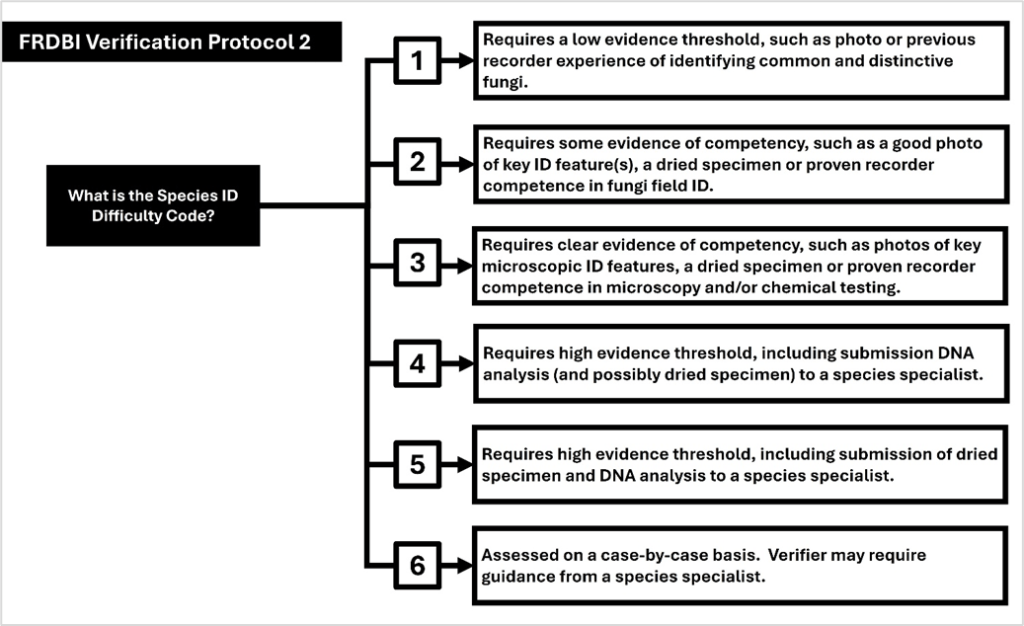
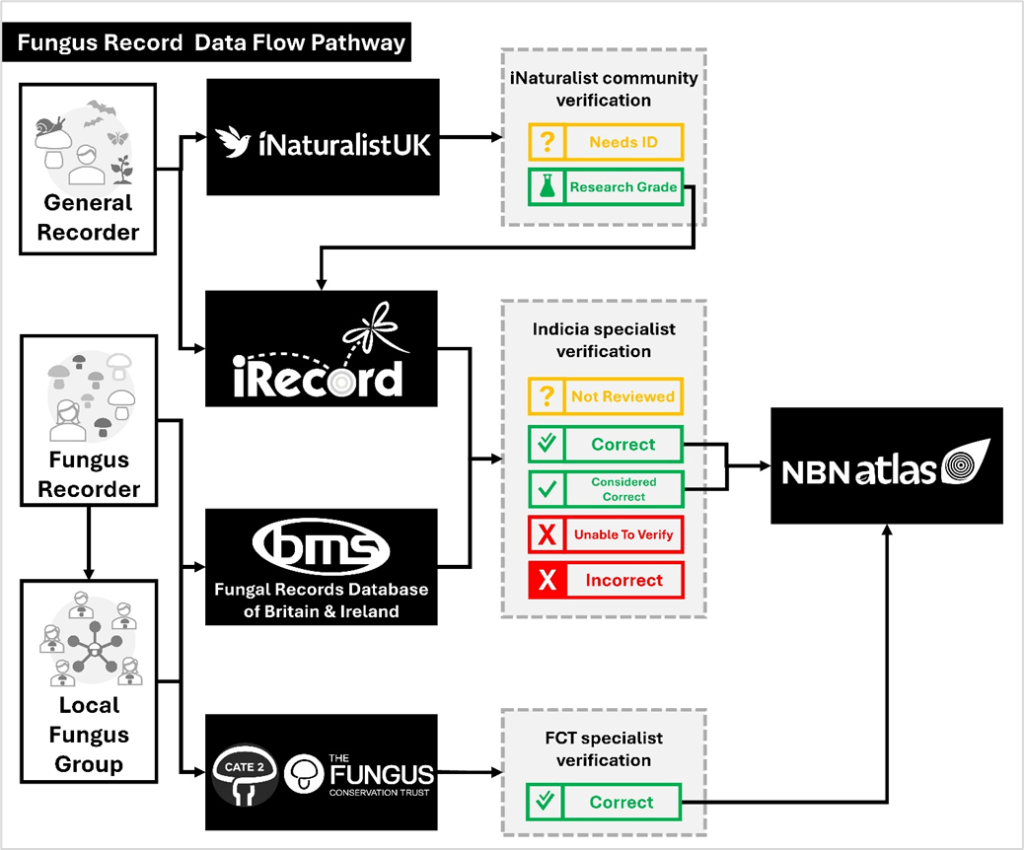
- Consultation projects include reviewing the pay of natural history tutors and working with the British Mycological Society to improve fungus record quality and data flow.