Sow Wild! and Insect Identification
Habitat loss and fragmentation are considered the foremost threats in pollinator decline, and in England and Wales, 97% of wildflower meadows were lost by 1984. Gardens have considerable potential for supporting pollinators, covering large areas of urban landscapes often with diverse floral resources. Buzz Club used citizen science to investigate the effectiveness of small 4m2 sown wildflower ‘mini-meadows’ in UK gardens and allotments in recruiting beneficial insects. They then examined effective methods and any potential bias in the identification and sampling methods conducted by citizen scientists. Janine will explain the results of the ‘Sow Wild!’ project, followed by Issy who will talk about the Buzz Club’s new ‘Insect ID Quiz’ project. The insect ID Quiz is helping the Buzz Club determine if training will help to limit any identification bias in citizen science observations.
Q&A with Dr Janine Griffiths-Lee
Dr Janine Griffiths-Lee is a post-doctoral researcher at RBG Kew. Based at Wakehurst, she works in the Nature Unlocked team studying pollinators in the landscape. Her current focus is the benefits of different tree species to pollinators, habitat management in urban gardens, and citizen science.
Does the richness of wild bee species refer to the number of species or the diversity of species?
This refers to the number of species. Looking at the diversity is more complicated and looking at the significance of this was not something that we were able to look at in detail. In the paper, we do discuss the diversity but it’s quite complicated, so if you are interested in the diversity of bees that we collected I’d recommend checking out the paper.
Is it possible to get details of the wildflower mixes used in the Sow Wild! Project?
All of our mixes are listed in the appendix of the research paper. I made my own mixes for this project, but the Buzz Club also have a really successful wildflower mix as part of our sponsor packs. Its lovely and bees come throughout the year with this mix.
You can find out more about becoming a sponsor and learn about our sponsor packs here: https://www.thebuzzclub.uk/memberandsponsor
Have you considered using bioacoustics and machine learning to look at insect activity in denser patches?
We are looking into different methods of bioacoustics and how accurate the level of detail is, for example, can you get to genus or just to broad groups? It was certainly out of scope for this study because it would have been a great expense to send such equipment to citizen scientists. We had 150 participants, so we really had to be quite aware of our limitations. But it is something interesting that I am working with at the moment – bioacoustics, and camera trapping as well.
Is it possible to get details of the wildflower mixes used in the Sow Wild! Project?
All of our mixes are listed in the appendix of the research paper. I made my own mixes for this project, but the Buzz Club also have a really successful wildflower mix as part of our sponsor packs. Its lovely and bees come throughout the year with this mix.
Q&A with Isobel Sexton
Issy Sexton is a Research Assistant at the University of Sussex, specialising in pollinator-focused citizen science. She also has a background in community engagement and environmental policy.
Are there any plans to follow on from the Sow Wild project?
Isobel: The Insect ID project leads directly on from it of course, but we don’t have any other projects in the pipeline leading on from this just yet. However, we’re always expanding our programme of projects so watch this space
Are the resources from these experiments available online?
The training webinar from the project is only available to the participants currently. The quizzes and online resources are free for everyone so you can go and try the quizzes here: https://www.thebuzzclub.uk/resources We also have flash cards. I haven’t created them yet for the bee project, but for others, you can print them off and take them into your garden to practise IDing. To access the training videos you need to be signed up as a project participant.
What is the plan for Insect ID in 2025?
What I’d really like to do is analyse the results and then take it forward in terms of how the skills gained from looking at the photos online are then transferred to the field. All of this is showing that it works really well online but how well does this translate when using the skills in the field?
I’d also like to look at the training and look at how long the skills are retained. Everyone was given two weeks to complete the first quiz, then a week’s break to do the training if they were in the training-between-quizzes group, or just a week’s break if they were in the control group, then they had two weeks to do the quiz. I left it like that because it was a citizen science project and I wanted people to enjoy doing it and not feel like it was a task. However, and again, I haven’t done the statistics on this yet, but just by looking, the people who did the training and then did the quiz straight away got the highest scores. Those who left it for two weeks still improved their scores somewhat. Those who did it straight away doubled their scores and the people who left it added four or five points. So that is something interesting to look at and to develop to make sure we can retain the information.
Literature References
- Klein et al (2007) Importance of pollinators in changing landscapes for world crops: https://doi.org/10.1098/rspb.2006.3721
- Ollerton et al (2011) How many flowering plants are pollinated by animals?: https://doi.org/10.1111/j.1600-0706.2010.18644.x
- Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services (2016) Summary for policy makers of the global assessment report of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services on pollinators, pollination, and food production: https://www.ipbes.net/assessment-reports/pollinators
- Baldock et al (2019) A systems approach reveals urban pollinator hotspots and conservation opportunities: https://doi.org/10.1038/s41559-018-0769-y
- Smith & Fellowes 2013 (2013) Towards a lawn without grass: the journey of the imperfect lawn and its analogues: https://doi.org/10.1080/14601176.2013.799314
- Goddard et al (2013) Why garden for wildlife? Social and ecological drivers, motivations and barriers for biodiversity management in residential landscapes: https://doi.org/10.1016/j.ecolecon.2012.07.016
- Balfour & Ratnieks (2022) The disproportionate value of ‘weeds’ to pollinators and biodiversity: https://doi.org/10.1111/1365-2664.14132
- Griffiths-Lee et al (2022). Sown mini-meadows increase pollinator diversity in gardens: https://doi.org/10.1007/s10841-022-00387-2
- Bonney et al (2009) Citizen Science: A Developing Tool for Expanding Science Knowledge and Scientific Literacy: https://doi.org/10.1525/bio.2009.59.11.9
- Birkin and Goulson (2015) Using citizen science to monitor pollination services: Citizen science and pollination services: https://doi.org/10.1111/een.12227
- Maher et al (2019) Using citizen science to examine the nesting ecology of ground-nesting bees: https://doi.org/10.1002/ecs2.2911
- Lye et al (2012) Using citizen science to monitor Bombus populations in the UK: Nesting ecology and relative abundance in the urban environment: https://doi.org/10.1007/s10841-011-9450-3
- Merenlender et al (2016) Evaluating environmental education, citizen science, and stewardship through naturalist programs: https://doi.org/10.1111/cobi.12737
- Gardiner et al (2012) Lessons from lady beetles: Accuracy of monitoring data from US and UK citizen-science programs: https://doi.org/10.1890/110185
- Burgess et al (2017) The science of 139 citizen science: Exploring barriers to use as a primary research tool: https://doi.org/10.1016/j.biocon.2016.05.014
- Law et al (2017) Crowdsourcing as a Tool for Research: Implications of Uncertainty: https://doi.org/10.1145/2998181.2998197
- Kremen et al (2011) Evaluating the Quality of CitizenScientist Data on Pollinator Communities: Citizen-Scientist Pollinator Monitoring: https://doi.org/10.1111/j.1523-1739.2011.01657.x
- Griffiths-Lee et al (2023) Sow Wild! Effective Methods and Identification Bias in Pollinator-Focused Experimental Citizen Science: https://doi.org/10.5334/cstp.550
Further Info
- Buzz Club website: https://www.thebuzzclub.uk/
- Insect ID Project page: https://www.thebuzzclub.uk/insectid
- Buzz Club Publications: https://www.thebuzzclub.uk/publications
- Buzz Club Quizzes: https://www.thebuzzclub.uk/resources
- Wildlife Gardening: https://www.wildlifetrusts.org/gardening
- Urban Food Agenda: https://www.fao.org/urban-agriculture/en/
- UK Pollinator Monitoring Scheme: https://ukpoms.org.uk/
- Wildlife Gardening Virtual Symposium: https://www.eventbrite.co.uk/e/wildlife-gardening-virtual-symposium-tickets-928434401287
- entoLEARN webinars: https://www.eventbrite.co.uk/cc/entolearn-webinars-1574569
entoLIVE
entoLIVE webinars feature guest invertebrate researchers delving into their own invertebrate research. All events are free to attend and are suitable for adults of all abilities – a passion for invertebrates is all that’s required!
- Donate to entoLIVE: https://www.gofundme.com/f/entolive-2025
- Upcoming entoLIVE webinars: https://www.eventbrite.com/cc/entolive-webinars-74679
- entoLIVE blog: https://biologicalrecording.co.uk/category/entolive-blog/
- entoLIVE on YouTube: https://youtube.com/playlist?list=PLuEBNUcfMmE95Re19nMKQ3iX8ZFRFgUAg&feature=shared
entoLIVE is delivered by the Biological Recording Company in partnership with the British Entomological & Natural History Society, Royal Entomological Society and Amateur Entomologists’ Society, with support from Buglife, Field Studies Council and National Biodiversity Network Trust.
The entoLIVE programme is delivered by Biological Recording Company and receives sponsorship from the following organisations:
- British Entomological & Natural History Society
- Royal Entomological Society
- Amateur Entomogists’ Society

More on citizen science
Protected: Earthworms of the UK Part 2
A World of Wasps: Researching the Ichneumonid Wasps
Huge numbers of insects are hosts to parasitoid wasps, and one of the most successful groups of wasps is the Ichneumonoidea, or Darwin Wasps. Despite the ubiquity of Darwin Wasps ,they are comparatively neglected by researchers and naturalists. This talk looks at research on species limits in these wasps and how that basic taxonomic work enables new research areas in ecology and genomics.
Q&A with Dr Gavin Broad
Dr Gavin Broad is a principal curator at the Natural History Museum, London, a taxonomist specialising on Ichneumonidae, and one of the co-investigators on Darwin Tree of Life, an ambitious project to sequence the genomes of all UK species.
Are ichneumonids all very host specific or do you have species that are more generalists?
They are often somewhere in the middle and it’s really hard to generalise. For example, within Netelia each species we know any detail about is probably going for a small range of hosts (though fairly closely related in the right habitat). Pimpla rufipes is a really common wasp that attacks all sorts of pupae that are the right sort of size. And not just Lepidoptera – they’ll sometimes go for other orders as well.
Does length of pupation impact which species different ichneumonids will target?
There are so many different factors. For example, you might get a species that has a six-month lifecycle and it needs two host generations a year. In that case, the potential host they could use in the autumn is really restricted to those that are going to overwinter in the correct stage. That’s a life history trait that can determine whether something’s a host or not. If they’re going to attack a caterpillar that then pupates late autumn, then this wasp will emerge in the winter when there’s nothing available for it.
What kinds of defences do the hosts have against these wasps?
There are lots of techniques. One thing that has been studied quite a bit are viruses that have been co-opted by wasps, enabling the wasp to overcome the host’s immune systembecause many insect larvae have quite robust immune systems that recognise foreign objects and encapsulate them. This results in a sort of arms race taking place inside the body of the host – as to how to stop the encapsulation by the host and being destroyed. There are also much simpler defences, if a Netelia puts its egg in the wrong bit of the caterpillar and the caterpillar can reach it with its mandibles, then it’s gone.
What do you need from the wasp in order to sequence it?
The tree within the presentation is based on barcode sequences so that’s a short 650 base pair fragment of mitochondrial DNA, cytochrome oxidase 1. We sequence that gene because it’s pretty informative at the species level. We usually extract the DNA from a leg of the specimen. For older specimens, we are still sequencing the same gene, but we have to use a very different technique to get the DNA in sufficient quantities because it is tiny and fragmented. We have to use a technique called genome skimming.
For genome sequencing, it is not as simple. The specimen has to be ultra fresh and we need to flash freeze it, using dry ice or a -80 degrees Celcius freezer. They freeze very quickly and they’re kept in deep cold until they are sequenced. At that point, the entire specimen is ground up and all of the available DNA is used. We may still sometimes need a second specimen for a DNA top-up. DNA does degrade quite rapidly and because we are using nanopore sequencing for everything, we need big, long strands of DNA for the whole genome. You’re pulling that DNA strand through a little pore in a film and so you want really big, long strands so that you don’t have too many little bits to knit together into a whole genome.
What percentage of parasitic wasps are ichneumonids and what percentage of these are nocturnal?
That’s a good question. In the UK, we’ve got roughly 6000 parasitoid wasps, and 2500 of those are ichneumonids. So, over a third. Not a lot of them are nocturnal. There are probably only around 150 truly nocturnal ones, not counting the ones that fly around at dusk.
Do we have a lot of species where we have little to no ecological behavioural information?
Yes, we don’t actually have any information on what the majority of Ichneumonids are doing. On the flipside, if you start rearing some interesting hosts, you’re almost guaranteed to find some sort of interesting parasitoid wasp and find a new host relationship. Studying Ichneumonids is frontier science!
How did you end up getting into taxonomy and why specifically the taxonomy of wasps?
Wasps was something I fell into. I wanted to be an ornithologist then I wanted to be a lepidopterist. And then I reared wasps, as an undergraduate, interesting parasitoids, and no one could really tell me what they were. I liked that challenge and decided to get into those. I found it fascinating because the whole biology of these things is amazing.
With taxonomy, I saw the word in a David Attenborough book when I was young, Living Planet or Life on Earth. One of those books that I had when I was too young to understand it really. But anyway, it mentioned taxonomists and I was intrigued. I always wanted to be a taxonomist and even when I found out what exactly it did involve, it didn’t put me off.
Literature References
- Broad & Shaw (2016) The British species of Enicospilus (Hymenoptera: Ichneumonidae: Ophioninae): https://europeanjournaloftaxonomy.eu/index.php/ejt/article/view/310
- Broad et al (2018) Ichneumonid Wasps (Hymenoptera: Ichneumonidae): their Classification and Biology: https://www.royensoc.co.uk/shop/publications/handbooks/hymenoptera/ichneumonid-wasps-hymenoptera-ichneumonidae-their-classification-and-biology/
- Broad et al. (2024) The genome sequence of an icheumonid wasp, Neteli virgata (Geoffroy 1785): https://wellcomeopenresearch.org/articles/9-187
- Johansson & Cedreberg (2019) Review of the Swedish species of Ophion (Hymenoptera: Ichneumonidae: Ophioninae), with the description of 18 new species and an illustrated key to Swedish species: https://europeanjournaloftaxonomy.eu/index.php/ejt/article/view/749
- Jonsonn et al (2021), A century of social wasp occupancy trends from natural history collections: spatiotemporal resolutions have little effect on model performance: https://nora.nerc.ac.uk/id/eprint/530080/1/N530080JA.pdf
Further Info
- Beginner’s Guide to Identifying British Ichneumonids (free download): https://www.nhm.ac.uk/content/dam/nhmwww/take-part/identify-nature/british-ichneumonid-wasps-id-guide.pdf
- Darwin Tree of Life project: https://www.darwintreeoflife.org/
- Unlocking Invertebrate Genomes entoLIVE blog: https://biologicalrecording.co.uk/2023/04/13/sequencing-invertebrates/
- Biodiversity Heritage Library: https://www.biodiversitylibrary.org/
- GBIF Netelia ornata: https://www.gbif.org/species/1281949
entoLIVE
entoLIVE webinars feature guest invertebrate researchers delving into their own invertebrate research. All events are free to attend and are suitable for adults of all abilities – a passion for invertebrates is all that’s required!
- Donate to entoLIVE: https://www.gofundme.com/f/entolive-2025
- Upcoming entoLIVE webinars: https://www.eventbrite.com/cc/entolive-webinars-74679
- entoLIVE blog: https://biologicalrecording.co.uk/category/entolive-blog/
- entoLIVE on YouTube: https://youtube.com/playlist?list=PLuEBNUcfMmE95Re19nMKQ3iX8ZFRFgUAg&feature=shared
entoLIVE is delivered by the Biological Recording Company in partnership with the British Entomological & Natural History Society, Royal Entomological Society and Amateur Entomologists’ Society, with support from Buglife, Field Studies Council and National Biodiversity Network Trust.
The entoLIVE programme is delivered by Biological Recording Company and receives sponsorship from the following organisations:
- British Entomological & Natural History Society
- Royal Entomological Society
- Amateur Entomogists’ Society

More on invertebrates
Can Passive Acoustic Monitoring of Birds Replace Site Surveys?
The technology used for monitoring the bioacoustics of a site is constantly evolving, with improved hardware, advancing software and growing use of Artificial Intelligence.
Are we heading towards a situation where using sound recorders to passively monitor a site for birds can replace the need for site surveys with human field surveyors?
This blog features presentations from a technology specialist and ecologist, exploring the benefits and limitations of undertaking passive acoustic monitoring of birds on a site. We delve into how these two methods can complement each other and hear case studies where passive acoustic monitoring has been used alongside traditional site surveys.
This blog features two guest presentations:
- Wilder Sensing: An Ecologist’s Perspective with Kate Downes (Ethos Environmental Planning)
- Wilder Sensing: Innovative Technology To Support Ecologists with Geoff Carss (Wilder Sensing)
Wilder Sensing: Innovative Technology To Support Ecologists
Geoff Carss (Wilder Sensing)
Wilder Sensing is a biodiversity monitoring and reporting tool that uses commercially available sound recorders to record soundscapes, processes the files using Artificial Intelligence to identify what species are present and provides easy-to-use analytic tools to understand the large datasets generated. This enables vast quantities of audio data to be captured and analysed at low cost to ecologists for both short and long-term surveys.
This presentation explores the practicalities and process of bioacoustics together with the results that can be achieved. Geoff Carss will explore some of the issues and limitations such as false positives and negatives, exclusion of under-reported species and bias depending on the species and how to mitigate them. He will also cover how to get long-term time series data sets, how monitoring in urban and industrial environments can cause bias due to background noise and how different filters can limit the identification of scarce species. This session will include a demo of Wilder Sensing where some of these issues will be explored.
Useful links
- Wilder Sensing: https://wildersensing.com/
- Contact Wilder Sensing: https://wildersensing.com/contact/
- How Can We Use Sound to Measure Biodiversity? Blog: https://biologicalrecording.co.uk/2024/07/09/bioacoustics-1/
- Springwatch 2024 Episode 11 (featuring acoustic monitoring): https://www.bbc.co.uk/iplayer/episode/m00205c3/springwatch-2024-episode-11
- Song Meter Micro 2: www.nhbs.com/song-meter-micro-2
Q&A with Geoff Carss
- Does Wilder Sensing only work for monitoring birds?
We’ve started with birds as we wanted to develop the model to work well for a single group before branching out. The tool can be used for other groups, but we first need to develop a model for dealing with the sounds created by these groups. The obvious next group would be bats and developing a model to deal with the ultrasonic calls made by various bat species. We’ve been undertaking work to investigate if the tool can be used for specific target species, such as Water Vole, and I’ve got some fantastic recordings of eels using hydrophones that we’re working on. There is also the fascinating emerging subject of soil acoustics, and other organisations looking at pollinator acoustics. The possibilities are vast, but in order for us to produce meaningful outputs we need to first understand the sounds made by various groups and then develop a model to recognise and interpret these. - Do you believe that, with further development, bioacoustics can be used to get measurements of abundance?
We’ve been recording on one particular site for over two years now. It is a site that has been “re-wetted” and the site managers are interested in how this has impacted the use of the site by Lapwings. We wanted to use the relative frequency of calls as a very rough approximation of abundance, with it being obvious when a flock of Lapwings arrive on the site. We hoped to be able to detect if more flocks were using the site for longer for winter feeding. However, this winter was so wet that the Lapwings were anywhere near the recorder due to the area being flooded out. A direct comparison using core frequency for abundance is pretty sketchy so we’re looking into the benefits of using triangulation, using multiple recorders to establish a measure of abundance. This will be quite the same as what a field surveyor would detect, but it’ll be interesting to see the results of using triangulation. - How much would it cost to get started?
We’ve deliberately set this up with a simple cost model. We charge a reduced rate of £400 per year for the analysis of data for one recorder for individuals, NGOs and charities. For ecological consultants and other for-profit businesses, we charge £800 per recorder per year. This includes covering the main cost associated with collecting this much data: the storage. We are constantly updating the tool. We don’t provide recorders, but you can use any recorder (such as the Song Meter Micro 2). We are always happy to chat over the Wilder Sensing tool and encourage anyone interested to get in touch.
Wilder Sensing: An Ecologist’s Perspective
Kate Downes (Ethos Environmental Planning)
Traditional field surveys are essential for understanding which species of bird are present on a site and how they are using it. However, these surveys only provide a snapshot in time and are limited by cost and field surveyor availability. Bioacoustic monitoring can monitor a site year-round but may miss things that field surveyors would detect. Kate discusses how Ethos Environmental Planning has developed a hybrid programme of bioacoustic and field surveys for undertaking ornithological site monitoring.
Kate Downes is an Ecologist at Ethos Environmental Planning and an Associate Member of the Chartered Institute of Ecology & Environmental Management. Kate’s specialism is ornithology, in her role at Ethos she leads the design and implementation of bird surveys across the seasons. Her previous work includes studying the Mauritius Kestrel as a field biologist and current work with barn owls helping the South Glos Owl and Bird of Prey Conservation Group (SGOBPC) monitor the species.
Useful links
- Ethos Environmental Planning: https://www.ethosep.co.uk/
- Get in touch with Kate: kate.downes@ethosep.co.uk
- Bird Survey & Assessment Steering Group (2024) Bird Survey Guidelines for assessing ecological impacts: https://birdsurveyguidelines.org
- Abrahams et al (2023) Good practice guidelines for long-term ecoacoustic monitoring in the UK: https://www.researchgate.net/publication/368683386_Good_practice_guidelines_for_long-term_ecoacoustic_monitoring_in_the_UK
- CIEEM Inpractice issue 125 (September 2024): https://cieem.net/i-am/in-practice/
Q&A with Kate Downes
- How does the bioacoustics approach compare financially to the traditional field survey approach?
That’s an interesting question, and not as straightforward to answer as you might think. We employed a hybrid approach, so we were using Wilder Sensing to complement our field surveys and this past year has very much been a trial for us. For smaller sites, it probably worked out cheaper as we were undertaking fewer field surveys and using the acoustics analysis to inform our reports. For the bigger sites, the recorders were an additional monitoring aspect on top of the usual field surveys and therefore were an additional expense, but gave us so much more data and enabled us to produce more robust reports. - Did you just use Wilder Sensing to analyse the acoustic data, or did you also undertake analysis?
We only used Wilder Sensing for the projects that I referenced in my talk. However, we did have a validation process for the data provided by Wilder Sensing. For example, if Wilder Sensing included a record of a Curlew at a site where this seemed unlikely we would check that using our own knowledge of as well as other bioacoustic analysis software. - How many recorders would you need for different habitats?
The guidance mentioned in the talk (see the links above) provides information on this and brings together all of the literature and experiments on this. There are lots of different things to consider here, including cost as each recorder and the data it produces has a cost associated with it. We set all of our recorders a minimum of 250 metres apart to reduce the chance of double counting as advised in the guidance.
Wilder Sensing ecoTECH blogs
- How Can We Use Sound to Measure Biodiversity: https://biologicalrecording.co.uk/2024/07/09/bioacoustics-1/
- Can Passive Acoustic Monitoring of Birds Replace Site Surveys blog: https://biologicalrecording.co.uk/2024/09/17/bioacoustics-2/
- The Wilder Sensing Guide to Mastering Bioacoustic Bird Surveys: https://biologicalrecording.co.uk/2024/11/26/bioacoustics-3/
- Bioacoustics for Regenerative Agriculture: https://biologicalrecording.co.uk/2025/03/31/bioacoustics-for-regen-ag/
- AI-powered Bioacoustics with BirdNET: https://biologicalrecording.co.uk/2025/07/08/birdnet/
- Making the Most of Bird Sounds: https://biologicalrecording.co.uk/2026/03/11/making-the-most-of-bird-sounds/
Event partners
This blog was produced by the Biological Recording Company in partnership with Wilder Sensing, Wildlife Acoustics and NHBS.
- Sign up for the Wilder Sensing e-newsletter: https://2e428x.share-eu1.hsforms.com/2XxP8d_6lRSmBIKH7uwruXQ
- Wildlife Acoustics Song Meter Micro 2: www.nhbs.com/song-meter-micro-2
- Wildlife Acoustics Song Meter SM5: https://www.nhbs.com/song-meter-sm5-acoustic-recorder
- Check out the NHBS Field Guide Sale: www.nhbs.com/spring-promotions

More for environmental professionals
Bees and Beyond: Ecosystem Services at a Garden Scale
Gardening success and invertebrate activity are deeply intertwined, for better or worse! Demonstrating that the beneficial side of this relationship vastly outweighs the negative is hugely important for maximising the ecological value of gardens – as well as improving horticultural outcomes. Pollination and pest control are the easiest of these ‘ecosystem services’ to illustrate at garden scales. These Buzz Club projects aim to highlight both in practical and entertaining ways.
Q&A with Dr Linda Birkin
Dr Linda Birkin is a specialist in citizen science and urban entomology. She has a particular interest in ecological outreach and wildlife gardening.
Do the bags that you used exclude all pollination, including wind pollination?
I used mesh bags and material so they kept the insects out but allowed wind and pollen to go through. I supplied the mesh material in the first year of the project and in the following two, volunteers provided their own mesh material, including a wedding veil and old net curtains!
Are there any plans to extend the Garden Shop project outside of the UK?
I have had some interest from people in other countries. It is a case of finding a collaborator in that country who can update the cost values. I send the spreadsheet with the cost values stripped out, with instructions to look up local values and put those in. The pollination requirement would still be the same.
Does pet poo provide any food for any invertebrates?
The thing about pet poop is that it depends on the pet. Cat and dog waste, for instance, is predator waste. They are predators that you wouldn’t have at these population numbers naturally and so there’s an unnaturally high level of nitrogen and phosphorous being deposited because of their poop. These shouldn’t be left lying around. Other pets that produce dung, like rabbits, are herbivores and you can compost their poop. It’s something that can go safely back into the environment.
There have been some interesting papers lately regarding dog poop and urine at nature sites and the difference in nutrient spikes in terms of over-fertilisation levels between the side of the path and further in. There are massive spikes in phosphorus and nitrogen just on the sides of the paths. Even if dogs are on a lead, they are still peeing on the side and that has a knock-on effect on the rest of the ecosystem.
There are invertebrates and other living organisms that use animal waste, but the issue is more about how quickly it degrades back into the environment and if it causes problems when it does. Dog and cat poo has quite a high level of bacteria and parasites – many that can cause problems for humans and that you wouldn’t find in that concentration naturally. Worming and flea treatments are another issue. They are designed to, for instance, to kill parasites in the animals but they are not designed to stop once they exit the animal. This is something we are also thinking about researching.
Literature References
- Birkin (2018) Pollination ecosystem services and the urban environment: https://hdl.handle.net/10779/uos.23455604.v1
- Birkin & Goulson (2015) Using citizen science to monitor pollination services: https://resjournals.onlinelibrary.wiley.com/doi/10.1111/een.12227
- Griffiths-Lee et al (2022) Sown mini-meadows increase pollinator diversity in gardens. https://doi.org/10.1007/s10841-022-00387-2
- Nicholls et a (2020). The contribution of small-scale food production in urban areas to the sustainable development goals: a review and case study: https://doi.org/10.1007/s11625-020-00792-z
- Burton et al (2024) Earthworm Watch: Insights into urban earthworm communities in the UK using citizen science: https://doi.org/10.1016/j.ejsobi.2024.103622.
- Gillner et al (2015) Role of street trees in mitigating effects of heat and drought at highly sealed urban sites: https://doi.org/10.1016/j.landurbplan.2015.06.005.
- Sheikh et al (2023) Efficacy of green infrastructure in reducing exposure to local, traffic-related sources of airborne particulate matter (PM): https://doi.org/10.1016/j.scitotenv.2023.166598.
Further Info
- The Buzz Club: https://www.thebuzzclub.uk/
- Bumblebees of the UK online course: https://courses.biologicalrecording.co.uk/courses/bumblebees
- Garden Shop Calculator: Garden Shop Calculator | Buzz Club (thebuzzclub.uk)
- Refurbishing the Bee Hotel: Different Bees, Different Needs entoLIVE blog article: https://biologicalrecording.co.uk/2024/04/11/refurbishing-the-bee-hotel/
- Keeping the Spark Alive: Volunteer Retention in Citizen Science blog article: https://biologicalrecording.co.uk/2024/05/16/volunteer-retention-in-citizen-science/
entoLIVE
entoLIVE webinars feature guest invertebrate researchers delving into their own invertebrate research. All events are free to attend and are suitable for adults of all abilities – a passion for invertebrates is all that’s required!
- Donate to entoLIVE: https://www.gofundme.com/f/entolive-2025
- Upcoming entoLIVE webinars: https://www.eventbrite.com/cc/entolive-webinars-74679
- entoLIVE blog: https://biologicalrecording.co.uk/category/entolive-blog/
- entoLIVE on YouTube: https://youtube.com/playlist?list=PLuEBNUcfMmE95Re19nMKQ3iX8ZFRFgUAg&feature=shared
entoLIVE is delivered by the Biological Recording Company in partnership with the British Entomological & Natural History Society, Royal Entomological Society and Amateur Entomologists’ Society, with support from Buglife, Field Studies Council and National Biodiversity Network Trust.
The entoLIVE programme is delivered by Biological Recording Company and receives sponsorship from the following organisations:
- British Entomological & Natural History Society
- Royal Entomological Society
- Amateur Entomogists’ Society

More on citizen science
Protected: Grove Farm Biological Recording Training
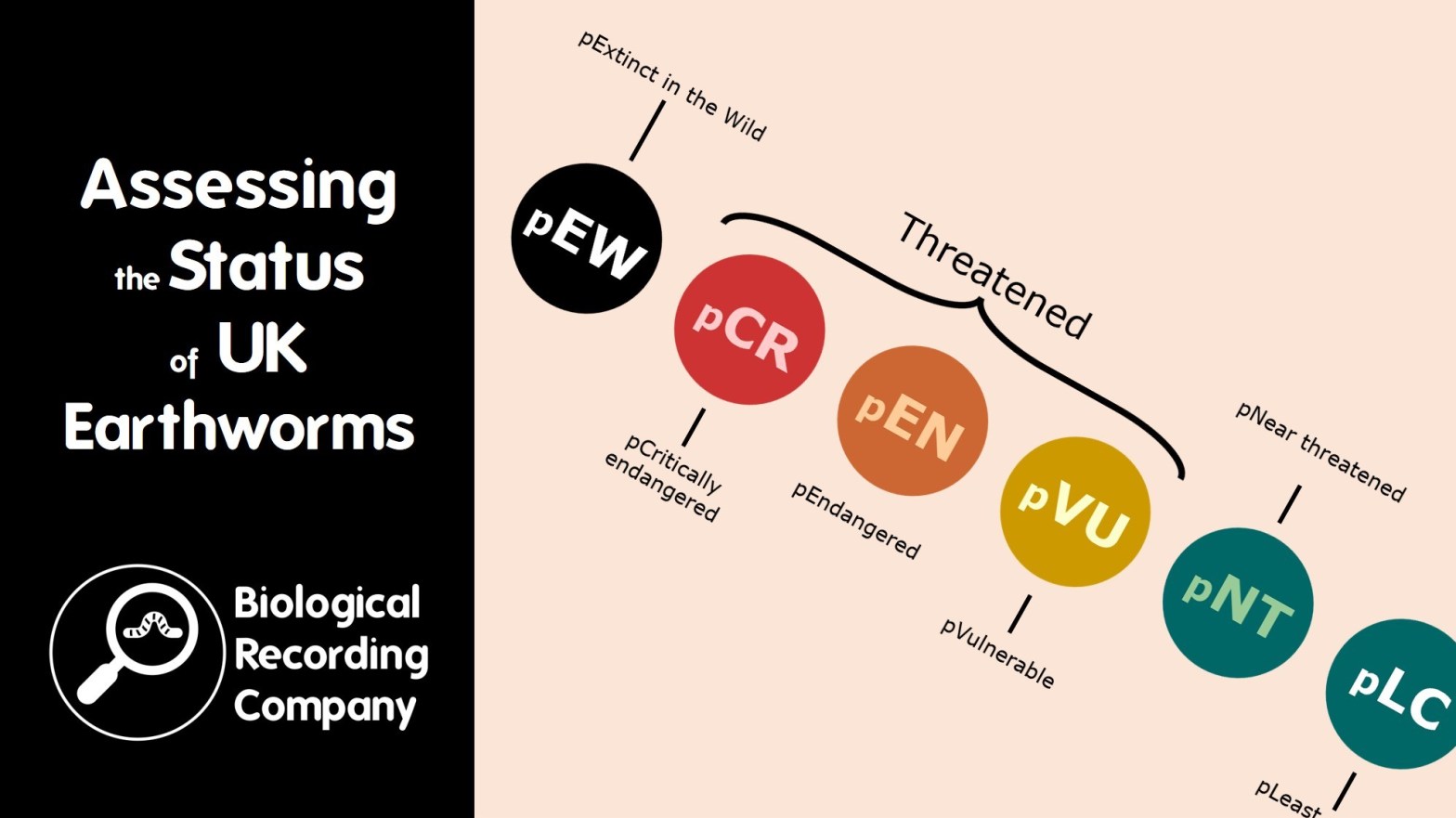
Assessing the Status of UK Earthworms
The standard method for determining the conservation status of a species is to assess the species against IUCN Red List criteria and apply an IUCN Red List category. These criteria require species data, and many species can therefore only be classed as ‘data deficient’ based on current record holdings. UK earthworms fall into this category and are rarely considered within site management plans, environmental policy or conservation projects.
The Provisional Earthworm Conservation Status Assessment for the UK aims to apply a provisional version of the IUCN categories for all 31 UK species of earthworm based on the little data that we do have and the opinions of the UK’s leading earthworm specialists. This presentation will explore this experimental approach and highlight the benefits and challenges resulting from this project.
Q&A with Keiron Derek Brown
Keiron Derek Brown has been running the National Earthworm Recording Scheme since it was launched in 2014. He has run over 100 earthworm training courses and events since 2014, covering a wide range of subjects including biology, ecology, surveying, identification and recording.
- How did you get involved with earthworm recording?
I personally became interested following volunteer work at the Natural History Museum, where I was trained to identify earthworms as part of a research project. When I then did some recording in Cumbria and sent the records to the Earthworm Society, I realised the data wasn’t being used or collated in any meaningful way so I volunteered to take on the role of Recording Officer and set up the National Earthworm Recording Scheme (NERS) in 2014. I’ve stuck with it because earthworms are so important ecologically and nobody else was doing this. I’m proud of the difference that I’ve been able to make due to the dirth of data that came before this. - How can others get involved with earthworm recording?
Earthworm identification does take a certain level of skill and use of a microscope. The Biological Recording Company runs Earthworm Identification courses aimed at beginners, but we understand that the fact that you need to collect, kill and preserve specimens plus access to a microscope is always going to limit the number of people willing to get involved. We also host regular Earthworm Sampling Days where we survey sites for earthworms and contribute data to the NERS database – all of which are free. - What is the optimum habit representation within the dataset?
When I speak about habitats being under-represented, what I really mean is that we have too little data on them. We’d expect more records from some habitats as they account for larger proportions of land use. For example, farmland accounts for 70% of the land in England. We need more information on the earthworm communities that inhabit different habitats, so we need to survey all habitats equally until we have that baseline data. Once we have better information across different habitats, the next step would be to address geographic representation (regardless of habitat). - Which species of earthworm are most at threat from development?
In general, earthworms are not at threat from development. Although development may impact species, for example if areas are concreted over, it is unlikely that this is enough to threaten extinction for most species (even locally). However, the earthworm species that are classed as very rare don’t have much data on where they do and do not occur. If a species has a very small number of locations where it occurs it is particularly at risk from local pressures, such as development, agricultural practices and even habitat management practices on nature reserves. I used development as an example of a potential threat to a species with a few isolated populations in the talk but there is no current known threat from development. - Is the classification of species as non/native or invasive based on incomplete information?
To clarify, none of the non-native species of earthworm found in the UK are classed as invasive as we aren’t aware of them causing harm to UK habitats or native species. Information on global distribution is severely lacking. It is even more difficult to establish the native range of a species because earthworms are moved around so much in soil and sold for fishing bait or compost. I’m only suggesting that 4 of the 31 species found within the UK are registered as non-native species, and this is because they belong to taxonomic groups known to originate from other continents. There are a number of other species that may or not be native – we need genetic studies to help us establish their relation to colonies elsewhere in the world and to establish where they originate from. - How would you sum up the status of earthworms in the UK?
Unknown. This report highlights that there is so much that we still don’t know. We still have big gaps in our knowledge of ecology, distribution and global context. We don’t even know what the natural ranges of many species is. Hopefully, this report will lead to some of these knowledge gaps being addressed so that we can conduct a full red listing exercise on UK earthworm species.
Literature References
- Ashwood et al (2024) Earthworm Records and Habitat Associations in the British Isles: https://doi.org/10.1016/j.ejsobi.2024.103642
- Carpenter et al (2012) Mapping of earthworm distribution for the British Isles and Eire highlights the under-recording of an ecologically important group: https://doi.org/10.1007/s10531-011-0194-x
- Jones & Eggleton (2014) Earthworms in England: distribution, abundance and habitats (NECR145): https://publications.naturalengland.org.uk/publication/5174957155811328
- Sims and Gerard (1999) Earthworms. Synopses of the British Fauna (New Series): https://www.field-studies-council.org/shop/publications/earthworms-synopsis/
- Sherlock (2018) Key to the earthworms of the UK and Ireland: https://www.field-studies-council.org/shop/publications/earthworms-aidgap/
- Bottinelli et al (2020) An explicit definition of earthworm ecological categories – Marcel Bouché’s triangle revisited: https://doi.org/10.1016/j.geoderma.2020.114361
- Capowiez et al (2024) Let earthworms be functional – Definition of new functional groups based on their bioturbation behaviour: https://doi.org/10.1016/j.soilbio.2023.109209
Further info and links
- National Earthworm Recording Scheme: https://www.earthwormsoc.org.uk/ners
- Wriggling Into Recording entoLIVE: https://biologicalrecording.co.uk/2023/02/02/earthworms/
- Digging Through the Dust: Rediscovering Historic Earthworm Records in Museums and Publications: https://youtu.be/Cw7MoK07yQs
- UK earthworm datasets on GBIF: https://www.gbif.org/publisher/0b8171d0-6b32-4ccc-bf3d-bf34b56c36d3
- Earthworm ecology: https://www.earthwormsoc.org.uk/earthworm-ecology
- IUCN Red List Categories & Criteria: https://www.iucnredlist.org/resources/categories-and-criteria
More for environmental professionals
Britain’s Endemic Invertebrates
Britain’s endemic invertebrates are the crown jewels of our biodiversity. There are 22 endemic species ranging from the Turk’s Earth Centipede (Nothogeophilus turki) to the Manx Shearwater Flea (Ceratophyllus fionnus). Last year Buglife undertook global IUCN Red List assessments for 20 of these species. This presentation will explain the assessment results and introduce participants to these amazing species.
Craig Macadam is Conservation Director with Buglife where he oversees all practical action and advocacy to restore invertebrate populations. He is the national recorder for mayflies, stoneflies and leeches, and represents Buglife on a number of fora including the IUCN Species Survival Commission and the IUCN Task Force on Global Freshwater Macroinvertebrate Sampling Protocols.
Q&A with Craig Macadam
- Are there many additional invertebrate species that are considered endemic to the British Isles rather than just Great Britain?
There are a few. Off the top of my head, I think there are a couple of beetles and a moth that are found in Ireland. I’ve been chatting to people in Ireland to enquire if there is a list of species that are endemic to Ireland so that we can cross reference this with our lists of UK species. - Has the Manx Shearwater Flea (Ceratophyllus fionnus) been searched for on the Calf of Man?
There have been very brief investigations of nest material at most of the Manx Shearwater (Puffinus puffinus) sites. However, there is a need to look at this in more detail, particularly in places like the Calf of Man, Skomer and other Manx Shearwater roosts. A lot of the existing work on this was done in the 1960s and 1970s, with the most recent data from the Isle of Rum. You would need a licence to take the material and we’re in discussions with the RSPB about how this could progress. - What is the current state of data for UK endemic invertebrate species?
It is a really mixed bag. For some species there is really regular monitoring, such as the Environment Agency monitoring of freshwater invertebrates. There are also some really active recording schemes that target some of these species, for example, Lee Knight is looking for the British Cave Shrimp (Niphargellus glenniei) in sites all over southwest England and he’s generated a great dataset. The active Dipterist Forum recording schemes involve surveys going on in areas where these species should be picked up if they are present so if they are not detected we can infer that they are absent. - How do you establish that a species is endemic and not present elsewhere?
It took a while! Every time I thought that I had my list completed, I’d receive emails from people asking about other species. We contacted experts across Europe and did a very extensive literature search, including looking at specialist journals for the various groups. Luckily, for some species there had been European or global reviews of the families which allowed us to get really up-to-date information. There are still other species on the long list that could be endemic but we don’t have enough information on them to state confidently that they are. - Are there any key actions that the public or conservationists can take to support our endemic invertebrates?
I’d advise people to support the efforts of invertebrate NGOs. At the moment we have the campaign to protect the Fonseca’s Seed Fly (Botanophila fonsecai) and save Coul Links from development, where over 11,000 people across Scotland and the UK have written to the Planning Minister urging him to call-in the application, resulting in a public inquiry. If you’re interested in a specific endemic species and record that group, keep an eye out for it and ensure that your data is making its way to the NBN Atlas.
Literature References
- Macadam (2023) Britain’s Endemic Invertebrates: https://cdn.buglife.org.uk/2023/11/Endemic-Species-Report-2023-Final.pdf
Further info and links
- Buglife: https://www.buglife.org.uk/
- IUCN: https://www.iucnredlist.org/en
- IUCN Red List Categories and Criteria: https://www.iucnredlist.org/resources/categories-and-criteria
- IUCN Red List Criteria summary sheet: https://www.iucnredlist.org/resources/summary-sheet
More for environmental professionals
Ten Years of Invertebrate Surveying in the Natural England Field Unit
This second iteration of the Natural England Field Unit (NEFU) will celebrate its tenth year in 2024. NEFU currently has 5 entomologists working on condition assessments for the organisation, as well as contributing to a host of other entomological work, including DNA work, supporting PhDs, training and a range of other work. Pete will introduce a range of this work, showing the valuable entomological work NEFU does.
Pete Boardman FRES has worked in the Natural England Field Unit (NEFU) since 2016, having previously run a number of training projects at the Field Studies Council for a decade, and freelanced for thirteen as an entomologist. He was until recently national recorder of the Cranefly Recording Scheme, and has extensively taught various entomological subjects for Natural England, Universities, Wildlife Trusts, etc.
Q&A with Pete Boardman
- Do the SNCOs for Northern Ireland, Scotland and Wales also have equivalent field units?
I don’t believe there are equivalent field units in Northern Ireland or Scotland. They will have funding to contract out services where needed, rather than delivering surveying in-house. In Wales, they have a team of desk-based staff that undertake surveys from existing data and aerial maps. We’re very aware that we are lucky to have this team in-house as a country agency, and it is worth reiterating that the NEFU doesn’t undertake all of our invertebrate site surveys and we still need to contract out these services once we’ve reached our capacity. - Are there any freshwater specialists in the NEFU?
We don’t have anybody within the field unit but always try to campaign for people with freshwater expertise as there are potential jobs that we could do if we had somebody with these skills. Currently, the freshwater work that NE undertakes would be contracted out to freshwater ecologist contractors. - Are you using any technologies to speed up the identification?
Most of our methods involve traditional identification using a microscope, particularly due to the complexities of trying to identify small invertebrates in the field. The large point of what we do with the Natural History Museum is to increase the databases so that in the future we are able to get a species list from DNA analysis of the malaise trap catch. - Do you undertake any identification in the field?
We are limited as to what we can do in the field because most of what we are looking at needs observation under a microscope. However, where we can identify a specimen in the field we will try to do so and release it once it is identified. We usually have an idea of which high-fidelity species we are likely to find on site so we can be on the lookout for these to limit the number of specimens that we take of these. As an entomologist becomes more experienced, the chances of them collecting rare specimens becomes lower as they have a better idea of what they’re looking for. - Can you impose a site designation if the landowner opposes it?
Natural England does have the power to do this. It can get quite expensive as it needs to go through the courts and we’d need to engage with legal experts.
Further info and links
- Pantheon invertebrate assemblages tool: https://pantheon.brc.ac.uk/
- iRecord: https://irecord.org.uk/
- Instagram: https://www.instagram.com/nefieldunit/
- Twitter/X: https://x.com/i/flow/login?redirect_after_login=%2Fnefieldunit
- Stratiomys singularior: https://species.nbnatlas.org/species/NBNSYS0000007840
- W114: https://pantheon.brc.ac.uk/lexicon/stream-river-margin
- W126: https://pantheon.brc.ac.uk/lexicon/seepage
- Glanville fritillary: https://butterfly-conservation.org/butterflies/glanville-fritillary